Instant access from all over the world
QCTMS TMF offers you a secure and centralized place where you can store documents of your electronic Trial Master File (eTMF). You do not need any further software. Your pre-installed web browser suffices to share documents instantly from all over the world.
Intuitive and smart
Most users of QCTMS TMF will only need a few minutes of training, because we have designed QCTMS TMF to be very intuitive. Nevertheless, you will find everything you expect from an eTMF: Uploading documents via e-mail or web-browser, audit trails for every document, tagging, deleting, restoring etc. Project assistants will work more productive by using favorite folders and smart copy functions. Search functions enable you so search your eTMF for keywords, editors etc.
Optional eISF available
Since 2017 QCTMS TMF optionally lets you maintain electronic Investigator Site Files (eISF). An eISF speeds up the process of distributing documents to your sites. Monitors can save precious time for on-site monitoring because they can check ISF completeness before actually visiting the site. It also lets you request read&understood confirmations for changes of important documents. This way you can easily track who already read a particular document.
In compliance with regulatory recommendations
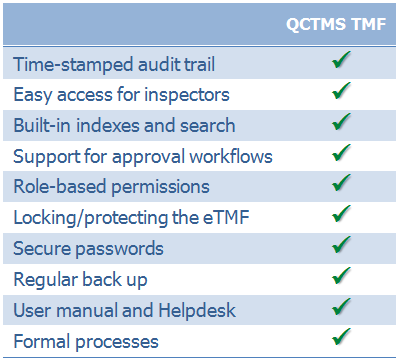
QCTMS TMF has been designed to comply with common standards and recommendations. We have summarized some of the requirements described in EMA’s reflection paper on GCP compliance related to TMF:
Andreas Grund and Jörg Mielebacher published an editorial on e-TMF in Journal for Clinical Studies 7 (4). It discusses the requirements and impacts. Please download it here.
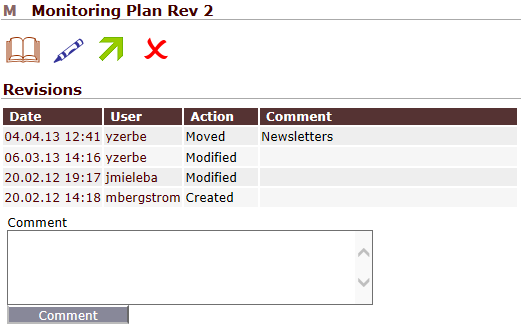
Storing documents in folders is not sufficient
One might think that storing TMF documents in the company’s file server is an appropriate eTMF. Besides many other drawbacks, this approach lacks one important feature: Such an eTMF is not audit-trailed. With QCTMS TMF you will always be able to see an automatically created audit trail for every document. Every previous revision of a document can be accessed easily. And of course, QCTMS TMF allows you to easily create folders.
Tailored and flexible
The electronic Trial Master File can be setup according to your SOPs. Folder hierarchies can be copied or moved with just a few clicks. Users and their roles can be added easily and in a timely manner.
Gaining insights
By using the power of QCTMS PM or RM, you will gain further insights in how your TMF looks like and how it is maintained. How frequent are document uploaded? How many empty folders are there? Are there any obviously corrupt files? How many documents are already approved?
When using QCTMS TMF together with QCTMS PM or RM, you will be able to analyze your TMF, e.g. frequency of document uploads, number of empty folders, and much more.